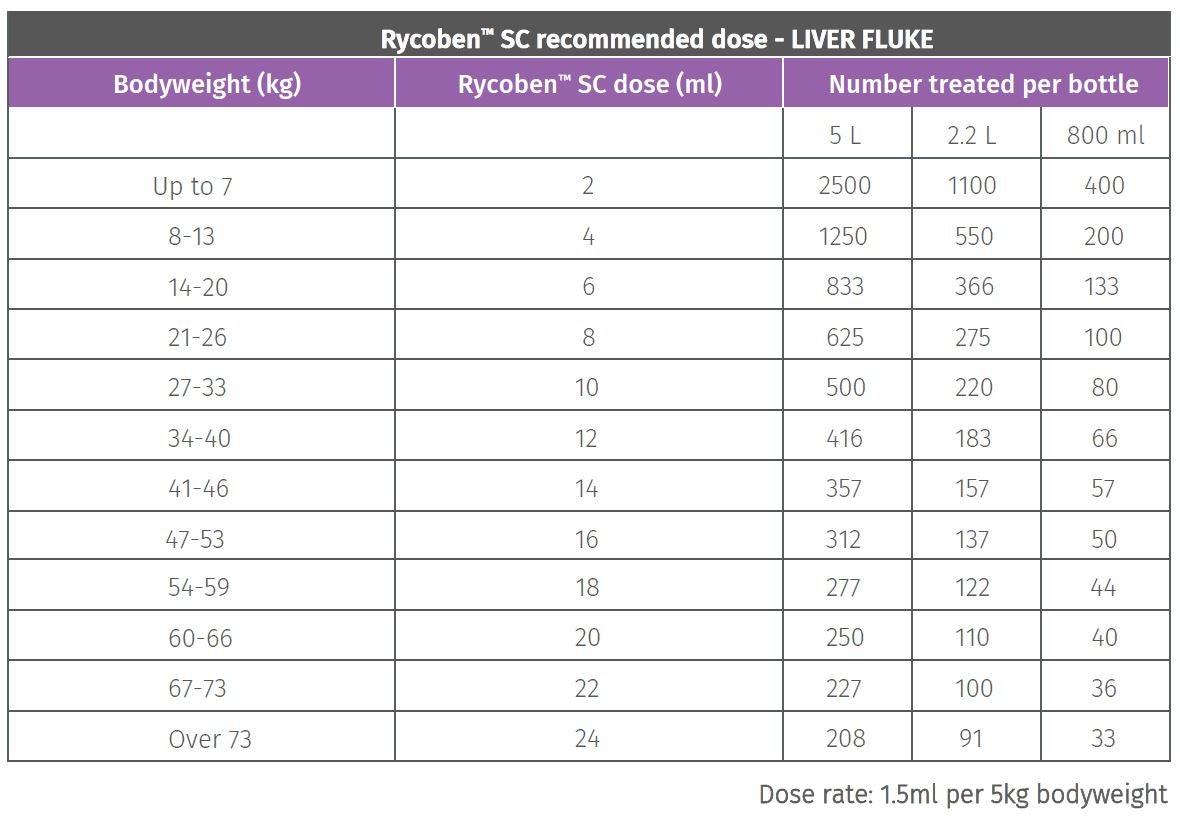
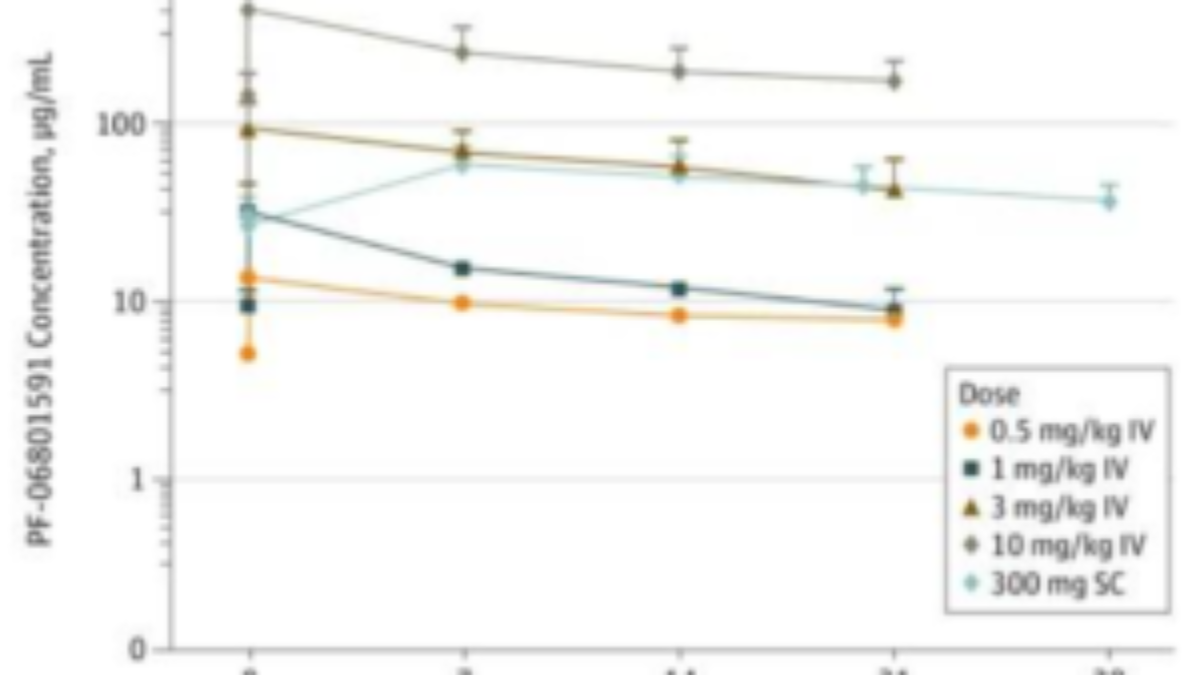
Dose-normalized plasma or serum concentration-time curves following... | Download Scientific Diagram
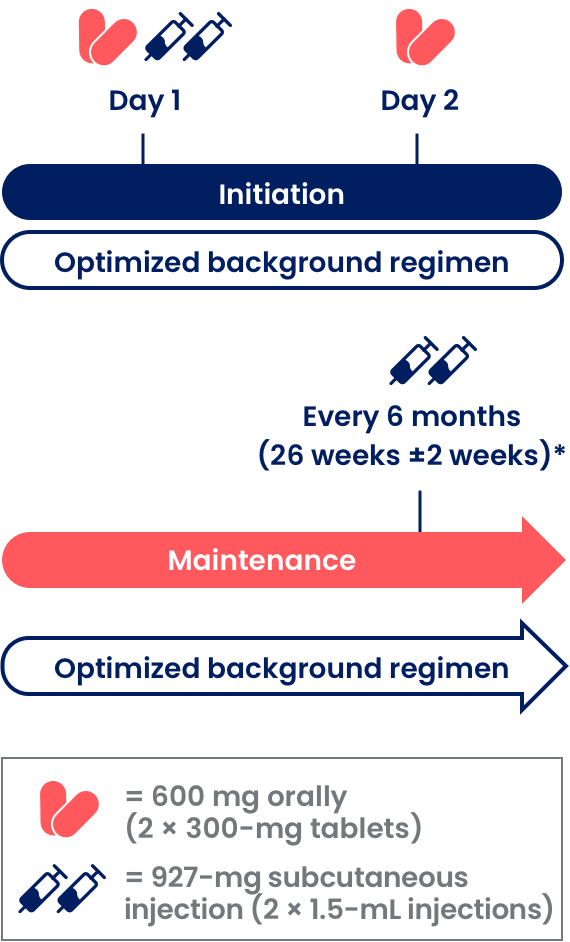
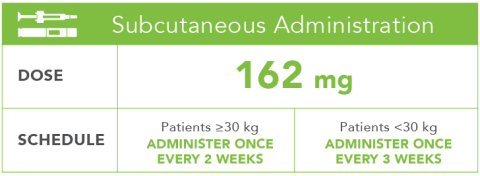
Large-Volume Subcutaneous Delivery: Clinical summary on the accuracy, safety, and efficacy of large-volume subcutaneous (SC) delivery
![PDF] Subcutaneous adjusted-dose unfractionated heparin vs fixed-dose low-molecular-weight heparin in the initial treatment of venous thromboembolism. | Semantic Scholar PDF] Subcutaneous adjusted-dose unfractionated heparin vs fixed-dose low-molecular-weight heparin in the initial treatment of venous thromboembolism. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/91b235faa3a1d8bd301c8387d81bade0a5cea3dd/2-Table1-1.png)
PDF] Subcutaneous adjusted-dose unfractionated heparin vs fixed-dose low-molecular-weight heparin in the initial treatment of venous thromboembolism. | Semantic Scholar

Comparison of intravenous and subcutaneous exposure supporting dose selection of subcutaneous belimumab systemic lupus erythematosus Phase 3 program - S Wickramaratne Senarath Yapa, D Roth, D Gordon, H Struemper, 2016

An intensified dosing schedule of subcutaneous methotrexate in patients with moderate to severe plaque-type psoriasis (METOP): a 52 week, multicentre, randomised, double-blind, placebo-controlled, phase 3 trial - The Lancet

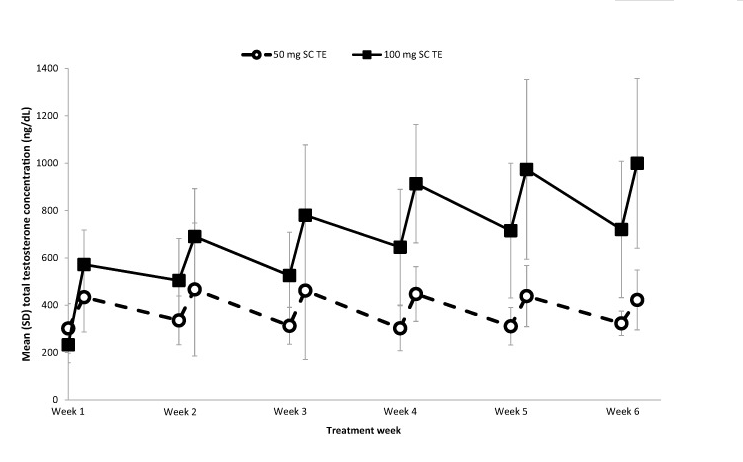
Pharmacokinetics and Safety of Subcutaneous Pasireotide and Intramuscular Pasireotide Long-acting Release in Chinese Male Healthy Volunteers: A Phase I, Single-center, Open-label, Randomized Study - ScienceDirect

Head-to-head, randomised, crossover study of oral versus subcutaneous methotrexate in patients with rheumatoid arthritis: drug-exposure limitations of oral methotrexate at doses ≥15 mg may be overcome with subcutaneous administration | Annals of