PPT - Human Specimen Repositories Requirements of 21 CFR Parts 50 & 56 PRIM & R May 5, 2004 PowerPoint Presentation - ID:3976095

Violations Of 21 CFR Part 56 - Institutional Review Boards: Warning Letters Issued by U.S. Food and Drug Administration (FDA Warning Letters Analysis): Chang, C: 9781514625903: Amazon.com: Books

Good Clinical Practice eRegs & Guides - For Your Reference Book 2 eBook by FDA - EPUB | Rakuten Kobo 1230000220467
Guidance on Special Considerations for the Oversight of Research Protocols using an FDA-Regulated Product

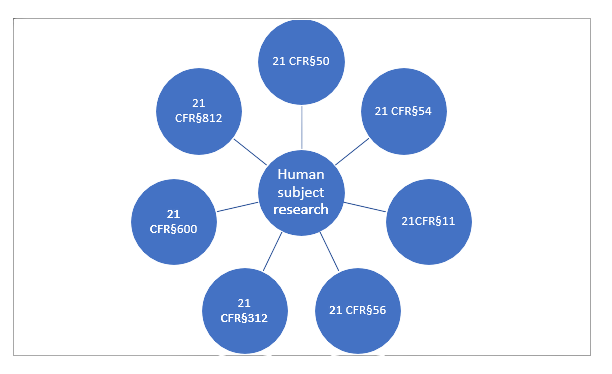
FDA Regulations (Part 1 & 2): 21 CFR Parts 312, 54, 50, 56, Form FDA 1572 & Investigator Responsibilities
Appendix 03 Links to Food and Drug Administration Code of Federal Regulations Protection of Human Subjects 21 CFR 50 Institu

PPT - FDA Regulations Pertaining to Good Clinical Practice and Clinical Trials PowerPoint Presentation - ID:474948

Compact Regs Parts 50, 54, 56, And 312 : CFR 21 Parts 50, 56, and 312 Good Clinical Practices (10 Pack) by Food and Drug Administration (2002, E-book) for sale online | eBay

Book M2: 2022 Mini Pocket-Sized (3" x 5") Code of Federal Regulations – Clinical Research Resources, LLC

Understanding Food and Drug Administration Regulatory Requirements for an Investigational Device Exemption for Sponsor-Investigators - M. E. Blair Holbein, Jelena Petrovic Berglund, 2012
Human Specimen Repositories Requirements of 21 CFR Parts 50 & 56 What I will be Covering FDA Human Research Protection Regul